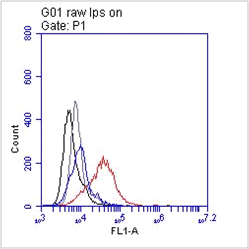
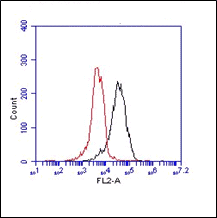
The monoclonal antibody B8E5 recognizes human muscarinic acetylcholine receptor M2 (M2 receptor), a G protein-coupled cardiovascular receptor of ~55 kDa. This receptor is an integral membrane protein consisting of seven membrane spanning a-helices linked together by extra- and intracellular loops that form a pharmacophore pocket. Autoantibodies directed against cardiovascular G protein-coupled receptors functionally interfering with the target have been described in several cardiovascular diseases. The M2 receptor is the predominant sybtype of muscarinic receptors present in the heart of mammalian species. The muscarinic acetylcholine receptor mediates various cellular responses, including inhibition of adenylate cyclase, breakdown of phosphoinositides and modulation of potassium channels through the action of G proteins. Primary transducing effect is adenylate cyclase inhibition. Monoclonal antibody B8E5 inhibits the ß-adrenergic L-type Ca2+ currents through activation of the muscarinic acetylcholine receptor M2. It suggests that the antibody acts not via the classical pathway of decreasing cAMP, but rather by increasing cGMP. Monoclonal antibody B8E5 acts by functional dimerization of the receptor resulting in stabilization of the constitutive active receptor dimers and paradoxically induces a small decrease in carbachol affinity for the M2 receptor. It recognizes the pentapeptide VRTVE (aa 168-172) corresponding to the N-terminal part of the second extracellular loop of the human M2 receptor.