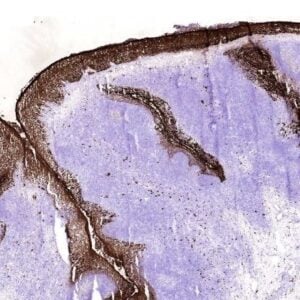
The polyclonal antibody recognizes the extracellular part of the human Tumor Necrosis Factor Receptor type 2 (TNF-RII) of the membrane-bound as well as the soluble receptor. TNF-RII (~75-80 kDa) is present on most cell types and is considered to play a prominent role in cell stimulation by TNF-alpha. TNF-alpha activates inflammatory responses, induces apoptosis, regulates cellular proliferation, and may even promote cancer progression. The effects of TNF-alpha are mediated by TNF-RI and TNF-RII, which have both distinct and overlapping downstream signaling cascades. Induction of cytotoxicity and other functions are mediated largely via TNF-RI. TNF-RI is equally well activated by both the 17 kDa soluble and 26 kDa membrane-bound form, whereas TNF-RII is efficiently activated only by the membrane bound form of TNF-alpha. Binding of the inherently trimeric TNF-alpha to TNFR1 and TNFR2 induces receptor trimerization and recruitment of several signaling proteins to the cytoplasmic domains of the receptors. Occupancy of TNFR2 results in direct recruitment of TNF Receptor Associated Factor 2 (TRAF2), which in turn recruits TRAF1.