The monoclonal antibody BV9 binds to the extracellular domain (EC3-EC4) of human VE-cadherin (vascular endothelial cadherin). Endothelial cells control the passage of plasma constituents and circulating cells from blood to the underlying tissues. VE-cadherin is of vital importance for the maintenance and control of endothelial cell contacts. Mechanisms that regulate VE-cadherinmediated adhesion are important for the control of vascular permeability and leukocyte extravasation. VE-cadherin regulates various cellular processes such as cell proliferation and apoptosis and modulates vascular endothelial growth factor receptor functions. Therefore, VE-cadherin is also essential during embryonic angiogenesis. The specialized function of VE-cadherin is lost or impaired in several pathological conditions – including inflammation, sepsis, ischemia and diabetes – which leads to severe, and sometimes fatal, organ dysfunction. Furthermore, abnormal increase in vascular permeability is often observed in pathological conditions, such as tumor-induced angiogenesis, macular degeneration, allergy, and brain stroke.
Endothelial permeability is regulated in part by the dynamic opening and closure of cell-cell adherent junctions. In vascular endothelium, adherent junctions are mainly composed of VE-cadherin, an adhesive receptor that is able to self-associate at endothelial cellcell contacts. VE-cadherin links endothelial cells together by homophilic interactions mediated by its extracellular part and associates intracellularly with the actin cytoskeleton via catenins. VE-cadherin belongs to the cadherin super-family of cellcell adhesion molecules, which are encoded by more than 200 genes in the human genome. Classical cadherins are Ca2+-dependent, homophilic, cell to cell adhesion molecules expressed in nearly all cells within solid tissues. Cadherins form a core adhesion complex that consists of a cadherin dimer, binding through its extracellular region to another dimer of cadherins expressed in adjacent cells, while its intracellular region is anchored to the plasma membrane and linked to the cytoskeleton. The VE-cadherin extracellular domain consists of five cadherin-type repeats, called EC (extracellular cadherin) domains that are bound together by calcium ions in a rod-like structure.
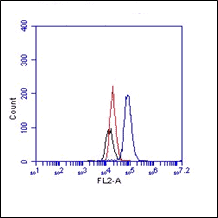
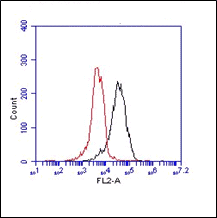
FC: Antibody BV9 stains the extracellular domain of VE-cadherin. As negative control an IgG isotype control was used (Ref.4)
FS: Antibody BV9 functions as an antagonist. The antibody was functionally tested by adding 10-50 µg/ml antibody BV9 to cell culture. The antibody blocks VE-cadherin causing a redistribution of VE-cadherin away from intracellular junctions(Ref.5, 6).
IA: Antibody BV9 can function as coat and detector.
IF: Cells on coverslips were fixed with 3% paraformaldehyde and permeabilized with 0.5% Triton X-100 before incubation with antibody BV9 (Ref.5, 8).
W: A reduced sample treatment and 7.5% SDS-Page was used. The band size is 130-140kDa (Ref.3).