Custom concentrations and volumes possible More info
TNF-RII, Mouse, mAb HM102
The monoclonal antibody HM102 recognizes the extracellular part of mouse membrane-bound TNF-RII as well as the soluble form of TNF-RII which is generated by proteolytic cleavage of the extracellular domain.
Read more€139.00 €9,220.00Price range: €139.00 through €9,220.00
The soluble form can still bind TNF-alpha with high affinity and functions as a TNF-alpha antagonist. TNF-alpha is an important signalling protein in the immune system which can activate inflammatory responses, induce apoptosis, regulate cellular proliferation, and may even promote cancer progression. TNF-alpha can bind to two structurally distinct membrane receptors, TNF-RI and TNFRII, which have both distinct and overlapping downstream signaling cascades. TNFRI is believed to be expressed on nearly all cell types, whereas TNFRII exhibits more restricted expression, being found on certain subpopulations of immune cells and several other cell types. A dominant role of TNFRII has been shown in thymocyte activation by TNF-alpha, whereas induction of cytotoxicity and other functions are mediated largely by TNF-RI. TNF-RI is equally well activated by both the 17 kDa soluble and 26 kDa membrane-bound form, whereas TNF-RII is activated only by the membrane bound form of TNF-alpha. The antibody is a agonistic receptor modulating antibody. It enhances in vitro TNF alpha responses by increasing the affinity of the soluble form of TNF-alpha for TNF-RII.
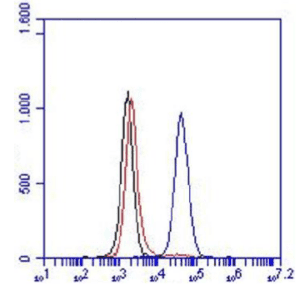
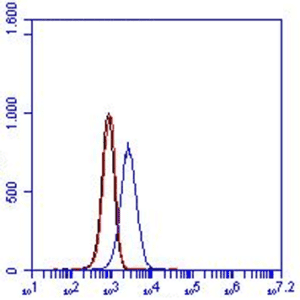
FC: 100.000 microglia cells were incubated with 10μg/ml PBS/1%serum for 30 min on ice
W: A reduced sample treatment and SDS-Page was used. The band size (s) is 75 kDa (Ref.4).
You may also like…
-
S100A9/Calprotectin, Mouse, ELISA kit
Cross reactivityGoat – Weak, Horse – No, Human – No, Pig – Weak, Rabbit – Weak, Rat – NoView product €929.00 €1,500.00Price range: €929.00 through €1,500.00 -
View product €715.00 €1,171.00Price range: €715.00 through €1,171.00
-
View product €715.00 €1,171.00Price range: €715.00 through €1,171.00
You may be interested in…
-
View product €139.00 €1,294.00Price range: €139.00 through €1,294.00
-
View product €139.00 €530.00Price range: €139.00 through €530.00
-
View product €139.00 €9,220.00Price range: €139.00 through €9,220.00
-
View product €139.00 €530.00Price range: €139.00 through €530.00