Custom concentrations and volumes possible More info
C3d, Human, clone HB2C2
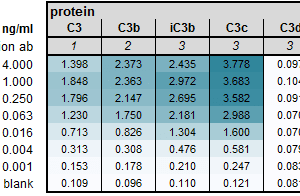
Antibody clone #HB2C2 recognizes a neo-epitope of complement C3dg. The antibody does not recognize C3 or (i)C3b.
Read more
€139.00 €474.00Price range: €139.00 through €474.00
The Vital Role of C3d in the Complement System and Immune Response
The complement system is a cornerstone of both innate and adaptive immune responses, offering protective and inflammatory reactions against pathogenic challenges before adaptive responses initiate. This article highlights the crucial role of C3d, a key fragment in this system.
Complex Mechanisms of the Complement System
This system is a complex array of proteins and receptors present in circulation, tissues, and other body fluids. It activates through three pathways: the classical pathway, triggered by immune complexes; the lectin pathway, initiated by surface-bound lectins; and the alternative pathway (AP), activated by surfaces not specifically protected against it. Each pathway creates a C3 convertase, cleaving the central complement protein C3 into C3b and generating C3d.
C3 Convertases: Catalysts in Complement Activation
C3 and C5 convertases are enzymatic complexes critical for initiating and amplifying complement pathway activities, leading to the formation of the cytolytic MAC. The synthesis of C3, from which C3d is derived, is tissue-specific and modulated by various stimuli. Post-cleavage, C3 yields anaphylotoxin C3a and activating fragment C3b.
The Path to C3d Formation
Once bound to the cell surface, C3b initiates the terminal complement pathway, forming the C5 convertase. Further cleavage of C3b by trypsin-like enzymes results in iC3b, subsequently leading to C3c and C3dg. Importantly, C3dg is further digested to form C3d, a process slower in blood, with C3dg being the predominant form.
C3d: A Key Player in Disease and Immune Regulation
C3, with a molecular weight of approximately 185kDa, is the most abundant protein in the complement system, with serum levels around 1.3 mg/ml. C3dg, and consequently C3d, play significant roles in diseases like transplantation rejection, kidney diseases, AMD, and various inflammatory diseases. Moreover, surface-bound C3 proteins, particularly through complement receptor2 (CR2), regulate the adaptive immune response.
C3d: A Biomarker for Immune-Related Diseases
Complement fragments, including C3d, are emerging as vital biomarkers for many diseases. The binding of C3dg to cell membranes, leading to the release of C3d, positions it as an attractive diagnostic biomarker, reflecting ongoing complement activation more accurately than C3 and C4 levels. However, most antibodies recognize an epitope on the C3d part of the alpha chain without differentiating between native and activated C3 proteins.
Not sure which C3 antibody to use?
With numerous options available, it is essential to select the right C3 antibody to ensure the success of your research. We designed a guide to assist you in making an informed decision:
Go to our C3 researcher’s guide and choose the right antibody
You may also like…
-
View product €786.00 €1,271.00Price range: €786.00 through €1,271.00
-
View product €786.00 €1,271.00Price range: €786.00 through €1,271.00
You may be interested in…
-
View product €139.00 €431.00Price range: €139.00 through €431.00
-
View product €139.00 €431.00Price range: €139.00 through €431.00
-
View product €139.00 €431.00Price range: €139.00 through €431.00
-
View product €139.00 €1,294.00Price range: €139.00 through €1,294.00