Custom controls available separately More info
C3d, Human, ELISA kit
C3d, a pivotal fragment of the complement protein C3, plays a crucial role in the body’s innate and adaptive immune responses.
Read more€929.00 €1,500.00Price range: €929.00 through €1,500.00
C3d, a critical fragment of complement protein C3, significantly impacts innate and adaptive immunity. This system, encompassing a diverse array of proteins and receptors in the body’s fluids and tissues, activates through three pathways: classical, lectin, and alternative. Each pathway generates a C3 convertase, splitting C3 into C3b and the anaphylatoxin C3a. C3b’s further breakdown on cell surfaces kickstarts the terminal complement pathway, creating C3d.
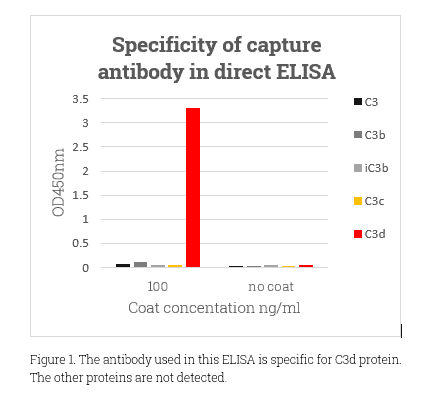
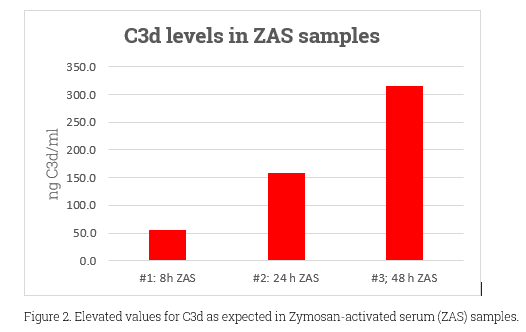
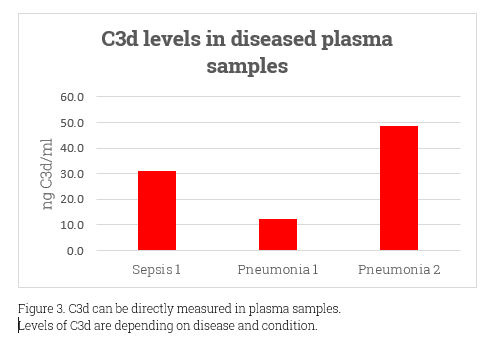
As the most abundant complement system protein, C3 has a molecular weight around 185kDa. C3d, at 38.9 KDa and non-glycosylated, emerges from C3b’s continual cleavage. It’s crucial in immune regulation, especially via interactions with complement receptor 2 (CR2). This role is essential in conditions like transplant rejection, kidney diseases, AMD, and various inflammatory diseases. C3d’s diagnostic value lies in its ability to indicate active complement activation, offering a more precise reflection than C3 and C4 levels. Despite the challenge that most antibodies can’t differentiate between C3d’s active and native states, its importance in the immune system and as a disease marker remains clear.
You may be interested in…
-
TCC, Human, ELISA kit
Cross reactivityCynomolgus monkey – Yes, Horse – No, Mouse – No, Pig – Yes, Rabbit – Yes, Rat – NoView product €786.00 €1,271.00Price range: €786.00 through €1,271.00 -
View product €786.00 €1,271.00Price range: €786.00 through €1,271.00
-
C3c, Human, ELISA kit
Cross reactivityCynomolgus monkey – Yes, Mouse – No, Pig – No, Rabbit – No, Rat – NoView product €858.00 €1,413.00Price range: €858.00 through €1,413.00 -
View product €786.00 €1,271.00Price range: €786.00 through €1,271.00