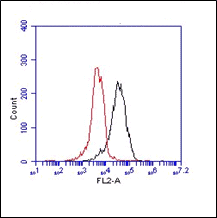
Monoclonal antibody HM2340, clone 1B1 recognizes human insulin growth factor 1 receptor. IGF-1R is a heterotetrameric transmembrane tyrosine kinase receptor expressed by many cells like lymphoid and myeloid cells as well as non haematopoietic cells. The receptor consists of two extracellular α subunits and two β subunits with tyrosine kinase activity. Receptor autophosphorylation follows by binding of its ligands insulin-like growth factor 1 and 2. The IGF/IGF-1R pathway plays important and diverse roles in tissue development and function, cell cycle progression, apoptosis and translation of proteins. IGF-1R regulates normal as well as pathological responses opening opportunities for therapeutic intervention. IGF-1R bears structural and functional resemblance to other tyrosine kinase receptors. Upon IGF binding the receptor is activated and autophosphorylated at the tyrosine residues in intracellular subunits leading to signaling via MAPK and PI3K/Akt pathways. Many tumors and transformed cells have altered IFG-1R expression. Recently the receptor came under the attention of roles in immune regulation. Proinflammatory cytokines, of which many share signaling components like Erk MAP kinases, seem to dampen several components of the IGF pathway. Furthermore, IGF regulates diverse aspects of T-cell, B-cell and monocyte function suggesting a role in pathogenesis of diseases where immunity is altered, like rheumatoid arthritis and IBD. Antibody 1B1 recognizes the extracellular domain if IGF-1R.