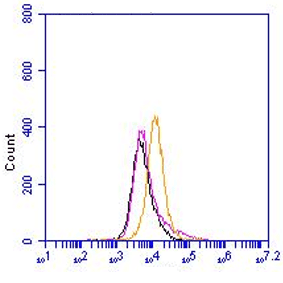
This polyclonal antibody recognizes the inner membrane protein SbmA of Gram-negative bacteria. The SbmA protein is a homodimeric secondary transporter protein, that is involved in the transport of microcins B17 and J25, glycopeptide antibiotic bleomycin, proline-rich antimicrobial peptides of prokaryotic and eukaryotic origin, antisense peptide phosphodiamidate morpholino oligomers and peptide nucleic acids (PNA) into the E.coli cytoplasm. This protein is found in a large number of Gram-negative and also Gram-positive microorganisms, including plant and animal pathogens. The monomeric SbmA protein contains 406 amino acids (about 50 kDa) and has 8 transmembrane regions. The homodimeric complex present in the membrane closely resembles the membrane-spanning region of the ATP-binding cassette transporter family. Peptide transport mediated by SmbA is driven by a electrochemical gradient. SbmA-mediated internalization of peptide substrates suggest that the transport of an unknown substrate(s) represents the function of this protein.