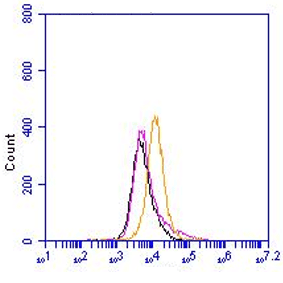
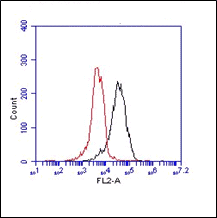
Mouse monoclonal antibody clone 3E2D10 recognizes human Von Willebrand Factor (vWF). vWF is a glycosylated protein expressed in endothelial cells (EC) and megakaryocytes. It contains multiple domains and exist as a multimer. vWF main function is to control blood hemostasis. Others functions include angiogenesis and vascular inflammation. It mediates platelet adhesion and stabilizes factor VIII. Its action is tightly regulated by ADAMTS13, which cleaves vWF at the peptide in domain A2. Majority of synthesized vWF multimers is stored in Weibel-Palade bodies of ECs or in platelets. The function of vWF is dependent on the size of the multimers. The larger the fragment, the stronger the thrombogenic capacity. Larger fragments bind more easily to circulating platelets. This underlines the importance of ADAMTS13 function. The vWF/ADAMTS13 axis is predominantly initiated under shear stress.
Von Willebrand disease (VWD) is the most common inherited bleeding disorder and is due to deficiency and/or abnormality of vWF. Measurement of vWF is key in the diagnosis of VWD. Nowadays, new roles are identified of the vWF/ADAMTS13 axis in vascular inflammation like process of leukocyte extravasation, ischemia injury, NETosis and complement activation. This implicates involvement in among others atherosclerosis, diabetes and sepsis. This make vWF an attractive biomarker in thrombinflammation.