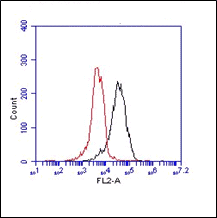
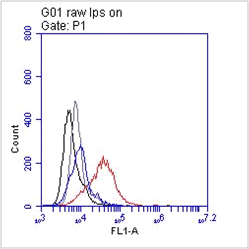
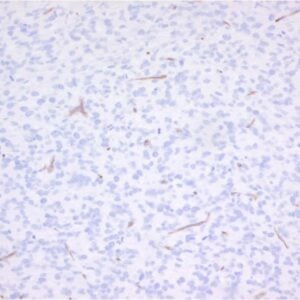
The monoclonal antibody TLR3.7 recognizes the 116 kDa human Toll-like receptor 3 (TLR3, CD283).
Toll-like receptors (TLRs) are highly conserved from Drosophila to humans and share structural and
functional similarities. TLRs constitute of a family of pattern recognition receptors (PRRs) that mediate
cellular responses to a large variety of pathogens (viruses, bacteria, and parasites) by specific
recognition of so-called ‘pathogen-associated molecular patterns’. Activation of TLRs, a family of at
least 11 different members that function either as homo- or heterodimers, leads to activation of NFκBdependent
and IFN-regulatory factor-dependent signaling pathways. TLRs have a central role in
innate immunity and are also required for the development of an adaptive immune response. TLRs
are expressed by various cells of the immune system, such as macrophages and dendritic cells. TLRs
are class I receptors, with a single α-helix that spans the cell membrane. They recognize and respond
to molecules derived from bacterial, viral and fungal pathogens, such as lipopolysaccharide (LPS)
from the outer membrane of Gram negative bacteria, peptidoglycan fragments from bacterial cell walls
and single-stranded and double-stranded RNA from viruses.
Some forms of RNA and DNA from pathogens exhibit immutable features that distinguish them from
nucleic acids of higher organisms. For example, dsRNA, is a common intermediate of viral replication
and a potent indicator of infection. Toll-like receptor 3 (TLR3) recognizes viral double-stranded RNA
and its synthetic analog polyriboinosinic:polyribocytidylic acid (poly(I:C)). TLR3 is normally located in
acidic endosomes where its luminal ectodomain (ECD) encounters dsRNA and induces type I
interferon (IFN), inflammatory cytokine/chemokine production and dendritic cell (DC) maturation via
the adaptor protein TICAM-1 (also called TRIF). Based on the different subcellular localization of
cytosolic RNA receptors and TLR3, these receptors seem to play distinct roles in anti-viral immune
responses.