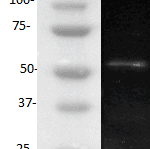
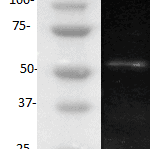
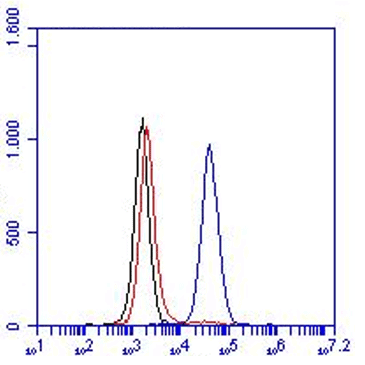
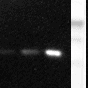
The monoclonal antibody H398 recognizes the extracellular part of the Tumor Necrosis Factor Receptor type I (TNF-RI) of the membrane-bound as well as the soluble receptor. TNF-RI (~55-60 kDa) is present on most cell types and is considered to play a prominent role in cell stimulation by TNF-alpha. TNF-alpha activates inflammatory responses, induces apoptosis, regulates cellular proliferation, and may even promote cancer progression. The effects of TNF-alpha are mediated by TNF-RI and TNF-RII, which have both distinct and overlapping downstream signaling cascades. Induction of cytotoxicity and other functions are mediated largely via TNF-RI. TNF-RI is equally well activated by both the 17 kDa soluble and 26 kDa membrane-bound form, whereas TNF-RII is efficiently activated only by the membrane bound form of TNF-alpha. TNF-RI signaling is initiated when trimeric TNF-alpha binds TNF-RI receptors. Subsequent TNF-RI trimerization promotes the recruitment of a proximal signaling complex composed of TNF Receptor Associated protein with a Death Domain (TRADD), Receptor Interacting Protein (RIP), cellular Inhibitor of Apoptosis Protein 1 (cIAP1), TNF Receptor Associated Factor 2 (TRAF2), and likely TRAF5. Studies with TNF-RI-deficient mice indicate that TNF-RI mediates most of the proliferation, pro-inflammatory, and apoptosis-activating pathways.
You may be interested in…
-
View product €139.00 €9,220.00Price range: €139.00 through €9,220.00
-
View product €139.00 €9,220.00Price range: €139.00 through €9,220.00
-
View product €139.00 €9,220.00Price range: €139.00 through €9,220.00
-
View product €139.00 €383.00Price range: €139.00 through €383.00