Custom controls available separately More info
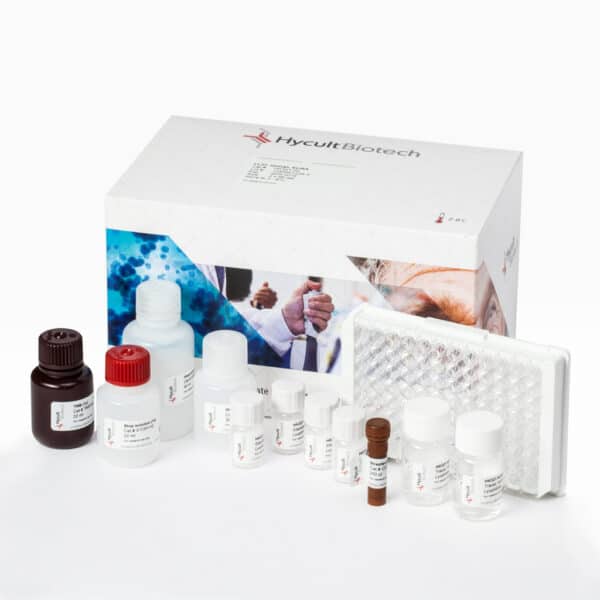
Complement factor B, Human, ELISA kit
In innate immunity’s arsenal, complement factor B stands as a vigilant defender, crucial for the alternative complement pathway’s activation and microbial destruction.
Read more€929.00 €1,500.00Price range: €929.00 through €1,500.00
Complement factor B (CFB), a key enzyme in innate immunity’s complement system, safeguards against microbial threats and has emerged as a crucial biomarker in clinical settings. Vital for the alternative complement pathway’s activation, CFB’s regulation is critical, particularly for protecting against encapsulated bacterial infections. Clinically, its deficiency links to increased susceptibility to infections such as; pneumococcal and meningococcal infections. Moreover, CFB’s diagnostic potential in pancreatic cancer is noteworthy; when combined with CA19-9 in the ComB-CAN panel, it significantly enhances early detection accuracy. Additionally, CFB may contribute to tumor growth by activating the PI3K-AKT pathway, making it an important focus for cancer screening and research.
You may also like…
-
View product €770.00
-
View product €858.00 €1,413.00Price range: €858.00 through €1,413.00
-
Properdin, Human, ELISA kit
Cross reactivityCynomolgus monkey – Yes, Goat – No, Horse – No, Mouse – Yes, Pig – Yes, Rabbit – Weak, Rat – No, Sheep – NoView product €858.00 €1,413.00Price range: €858.00 through €1,413.00 -
View product €929.00 €1,500.00Price range: €929.00 through €1,500.00