The monoclonal antibody 1-1C12 recognizes free LL-37 as well as its precursor human CAP18 (hCAP18). hCAP18 is a cathelicidin protein of ~18 kDa. It belongs to the cathelicidin family of antimicrobial peptides that are characterized by a conserved N-terminal cathelin domain and a variable C-terminal antimicrobial domain. Cathelicidins are predominantly found in the peroxidase-negative granules of neutrophils. During bacterial infections, the life span of neutrophils is regulated by various pathogen- and host-derived substances. Within the neutrophils, the cathelicidins are synthesized as preproproteins. After removal of the signal peptide,they are stored in granules as inactive proforms. The C-terminal antimicrobial peptides are activated when cleaved from the proforms of the cathelicidins by serine proteases from azurophil granules.
hCAP18 is a major protein in specific granules of neutrophils, but it is also present in subpopulations of lymphocytes and monocytes, in squamous epithelia (of the mouth, tongue, esophagus, cervix, and vagina), pulmonary epithelium, keratinocytes in inflammatory skin diseases and in the epididymus. The antibacterial C-terminus of hCAP18, LL-37 (37 amino acids), has been shown to exert broad antimicrobial activity towards gram-negative as well as gram-positive bacteria and to have synergistic antibacterial effects with the defensins. For instance deficiency in saliva, LL-37 accords with occurrence of periodontal disease in patients with morbus Kostmann. LL-37 does not only kill bacteria, but can also modulate (suppress) neutrophil apoptosis via the activation of FPRL1 and P2X7 in bacterial infections. Suppression of neutrophil apoptosis results in the prolongation of their life span, and may be advantageous for host defense against bacterial invasion. Moreover it functions as a chemotactic agent for neutrophils, monocytes and T cells. LL-37 is markedly resistant to proteolytic degradation and to a limited extent also cytotoxic towards mammalian cells.
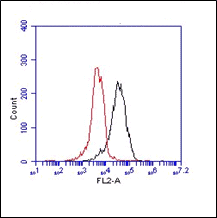
IF: paraformaldehyde fixed cells were incubated with 1-1c12 (1:50 diluted)
P: Sections were deparaffinized with sodium citrate and stained for LL-37 (1:50 diluted)
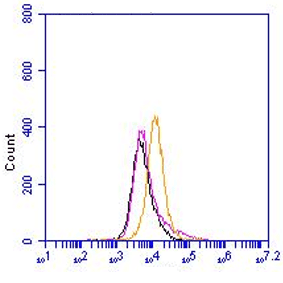
W:- A non-reduced sample treatment and denaturing or native SDS-PAGE was applied. Proteins were transferred to nitrocellulose, blocked with 5% dry milk and incubated with 1-1c12 , 1:250-1000 dilution. The band sizes are 18 and 4.5 kDa for hCAP-18 and LL-37 respectively (Ref.1-3)