Custom concentrations and volumes possible More info
Alpha-1-antitrypsin, Human, mAb 2C1
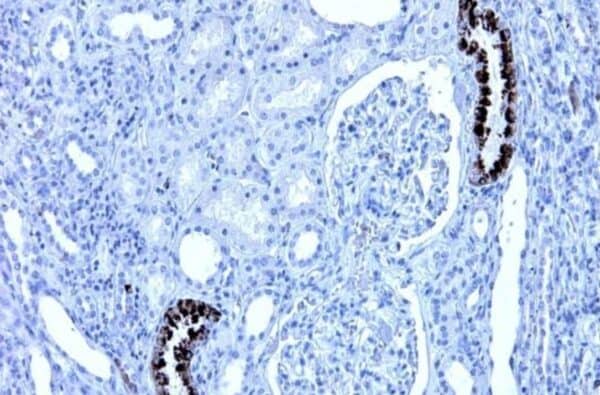
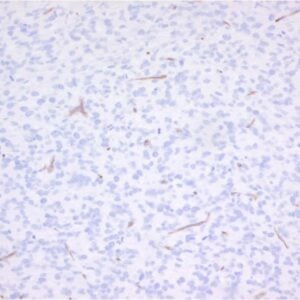
The mouse monoclonal antibody clone 2C1 recognizes polymeric forms of human alpha-1-antitrypsin.
Read more€139.00 €1,294.00Price range: €139.00 through €1,294.00
The mouse monoclonal antibody clone 2C1 recognizes polymeric forms of human alpha-1-antitrypsin.
Alpha-1-antitrypsin is the most abundant circulating protease inhibitor. Serpinopathies are conformational diseases characterized by the polymerization and intracellular retention of members of the serine protease inhibitor or serpin superfamily of proteins.1
The best known is a1-antitrypsin deficiency, with the most common severe deficiency allele being the Z mutation (Glu342Lys). The severe Z deficiency allele (Glu342Lys) causes the protein to undergo a conformational transition and form ordered polymers that are retained within hepatocytes. This causes neonatal hepatitis, cirrhosis, and hepatocellular carcinoma.
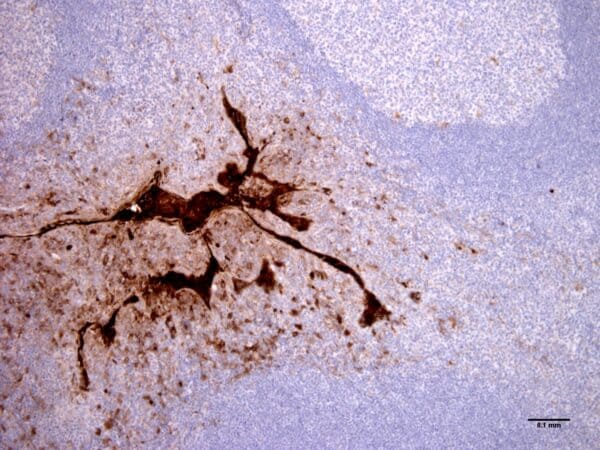
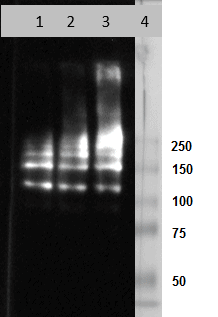
Clone 2C1 recognizes polymers formed by Z α1-antitrypsin in vivo. It also recognizes polymers formed by the Siiyama (Ser53Phe) and Brescia (Gly225Arg) mutants, and the novel His334Asp shutter domain mutant of a1-antitrypsin that is associated with prolonged neonatal jaundice in a 6-week-old boy. These data show that Z and shutter domain mutants form polymers with a shared epitope.
IA: HM2289 can be used as capture and detection antibody.