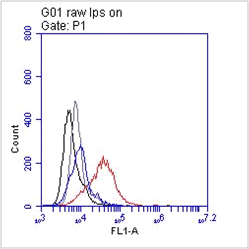
Antimicrobial proteins (AMP) are a fundamental element of the primary response against pathogens. AMP’s are small endogenous cationic molecules expressed by phagocytic and epithelial cells. The antimicrobial activity of AMP’s is directed towards a broad spectrum of pathogens, like Gram-positive & negative bacteria, viruses, yeast and fungi. AMP’s aid in innate and adaptive immunity via direct inactivation and by immunomodulatory activity like leukocyte migration. Defensins are the most prominent mammalian AMP’s. Three defensin peptide families are identified, the α-, β-, and θ-defensins. They are characterized by a triple-stranded β-hairpin structure, six disulfide-linked cysteine residues and a positive charge. They are synthesized as preproteins and undergo processing to become a fully active peptide. Defensins are divided in alpha- and beta-defensins depending on their disulfide bridging pattern. α- and β-defensins modify cell migration and maturation, induce cytokines and trigger histamin and prostaglandin D2 release from mast cells. There are six α-defensins: HNP (human neutrophil peptide)-1 to -4, HD5 & 6. Human beta-defensin 1(hBD-1) is considered a prominent AMP in epithelial defense against infection. High expression levels have been found in the kidney and female reproductive tract, especially in pregnant women. Besides antimicrobial activity, hBD1 has an effect on chemoattractive immature dendritic cells and probably memory T cells. After reduction of disulphide-bridges hBD-1 becomes a potent antimicrobial peptide against the opportunistic pathogenic fungus Candida albicans and against anaerobic Gram positive commensals of Bifidobacterium and Lactobacillus species.