C3c, Human, mAb 4
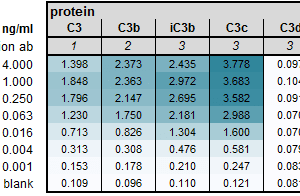
The monoclonal antibody 4 recognizes the cleaved human C3 fragment C3c. It recognizes a conformational epitope in C3c, C3b and iC3b.
Read more€133.00 – €414.00
Understanding the Role of C3c in the Complement System and Immune Response
The complement system is critical in both innate and adaptive immune responses, producing inflammatory and protective reactions against pathogenic challenges. This article sheds light on the role of C3c, a vital component of this system.
The Complement System: A Network of Proteins and Receptors
Comprising a complex family of proteins and receptors, the complement system operates in circulation, tissues, and various body fluids. It activates through three pathways: the classical pathway, initiated by immune complexes; the lectin pathway, triggered by surface-bound lectins; and the alternative pathway (AP), activated by unprotected surfaces. Each pathway generates a C3 convertase, a serine protease that cleaves the central complement protein C3 into C3b.
C3 Convertases: Amplifiers of Complement Activity
C3 and C5 convertases are crucial in initiating and amplifying the activity of the complement pathways, leading to the formation of the cytolytic MAC. The synthesis of C3, and consequently C3c, is tissue-specific and modulated in response to various stimuli. The cleavage of C3 by C3 convertase results in the formation of C3a and C3b.
The Formation and Significance of C3c
Upon binding to the cell surface, C3b initiates the terminal complement pathway, forming the C5 convertase. Further cleavage by trypsin-like enzymes transforms C3b into iC3b, then C3c, and finally C3dg. The slow transformation of C3dg into C3d in the blood means that C3dg is more prevalent. C3, with a molecular weight of approximately 185 kDa, is the complement system’s most abundant protein, with serum levels around 1.3 mg/ml.
C3c: A Biomarker in Disease Diagnosis
C3 activation products, including C3c, play roles in various diseases such as transplantation rejection, kidney diseases, AMD, and inflammatory diseases. Surface-bound C3 proteins, particularly through complement receptor 2 (CR2), are instrumental in regulating the adaptive immune response. Consequently, complement fragments like C3c serve as biomarkers for many of these conditions. While C3dg’s binding to the cell membrane is unstable, its release makes it an attractive diagnostic biomarker, reflecting ongoing complement activation more accurately than C3 and C4 levels.
Not sure which C3 antibody to use?
With numerous options available, it is essential to select the right C3 antibody to ensure the success of your research. We designed a guide to assist you in making an informed decision:
Go to our C3 researcher’s guide and choose the right antibody
You may also like…
-
C3c, Human, ELISA kit
Cross reactivityCynomolgus monkey – Yes, Mouse – No, Pig – No, Rabbit – No, Rat – NoView product €825.00 – €1,359.00 -
View product €894.00 – €1,443.00
-
C3d, Human, ELISA kit
Cross reactivityCynomolgus monkey – Yes, Horse – No, Mouse – No, Pig – No, Rat – NoView product €894.00 – €1,443.00
You may be interested in…
-
View product €133.00 – €414.00
-
View product €133.00 – €414.00
-
View product €133.00 – €456.00