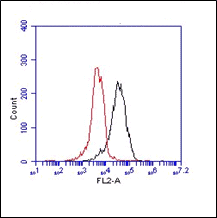
The monoclonal antibody MBI-1 recognizes Complement Factor I (CFI). CFI is a protein of the complement system (serine protease) , also known as as C3b/C4f inactivator, and is a protein that is encoded by the CFI gene, located on chromosome 4. It regulates complement activation by cleaving cell bound or fluid phase C3b and C4b. CFI is synthesized mostly in the liver, and is initially secreted as a single 88 kDa gene product; this precursor protein is then cleaved by furin to yield the mature CFI protein, which is a disulfide-linked dimer of heavy chain residues (residue 19-335, 51 kDa) and light chain (residues 340-583, 37 kDa). Only the mature protein is active. genetic polymorphism in CFI has been observed (variants R201S, R406H, R502L). CFI deficiency leads to low levels of complement component 3 (C3) in plasma, due to unregulated activation of the complement alternative pathway, and it has been associated with recurrent bacterial infections in children; more recently, mutations in the CFI gene have been shown to be implicated in development of Haemolytic Uremic Syndrome, a renal disease also caused by unregulated complement activation. The monoclonal antibody can be used to detect CFI levels in serum, plasma.