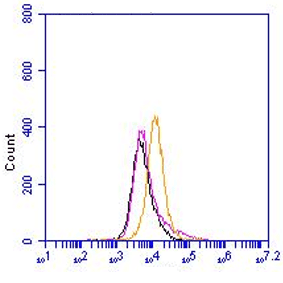
Monoclonal antibody clone 5F12.1.2, anti bovine Lactoferricin B is highly specific for bovine Lactoferricin B. This peptide is derived by enzymatic cleavage of lactoferrin which is a member of the transferrin family of metal-binding proteins found in milk and other secretory fluids and also in blood. Cleavage by pepsin of bovine lactoferrin leads to the release of Lactoferricin B (aminoacid 17-41). This peptide is highly basic, possessing five Arg (R) and three Lys (K) residues. In addition, a number of Trp (W) and Phe (F) aromatic residues are present. The two Cys (C) residues from lactoferricin B form a disulfide bond, generating an almost completely cyclical peptide. Nevertheless, the disulfide bond is not required for the antimicrobial potency. Several studies have shown that Lactoferricin B has a broad-spectrum activity against various Gram-positive and Gram-negative bacteria. In addition the peptide has been shown to have antifungal, antiviral and antitumour activity and to bind lipopolysaccharides (LPS, endotoxin). Moreover, it is known to stimulate the adaptive immune response and has anti-inflammatory properties. Lactoferricin B belongs to a large group of cationic antimicrobial peptides. The monoclonal antibody 5F12.1.2 is specific for bovine Lactoferricin B and detects the QWR antigenic determinant specific for bovine Lactoferricin B (3kDa), it lacks reactivity with bovine lactoferrin C-lobe, human lactoferrin or lactoferricin H. The QWR sequence recognized by the antibody 5F12.1.2 is not present in lactoferrin in human, pig, mouse, goat, rabbit, horse, rat, cockroach and African clawed frog.