Monoclonal antibody ER-TR7 recognizes with an intracellular component of mouse fibroblasts. Fibroblasts are the least specialized cells in the connective-tissue family. They are dispersed in connective tissue throughout the body, where they secrete a nonrigid extracellular matrix (ECM) that is rich in type I and/or type III collagen. Conective tissue consists of glycosaminoglycans, proteoglycans and glycoproteins through which various fibres run. These fibres can be collagenous, elastic or reticular. Reticular fibres are composed from the family of collagen proteins and give tensile strength. These fibres are made by reticular fibroblasts. The activation of fibroblasts by inflammatory stimuli results in their migration, proliferation and deposition of extracellular matrix components, important features involved in both wound healing and fibrosis.
The ER-TR7 antigen is a ubiquitous component of stromal (interstitial) matrix cartilage and of at least some basement membrane zones. The antigen detected is not a basement membrane component, nor any major collagen type or fibronectin. The antigen detected has a wider tissue distribution than reticulin. ER-TR7 detects an intracellular component of fibroblasts. Since ER-TR7 does not react with purified laminin, collagen types I-V, fibronectin, heparin sulfate proteoglycan, entactin or nidogen, it detects a hitherto uncharacterized antigen.
The monoclonal antibody ER-TR7 can be used to study the micro-anatomy of various organs. ER-TR7 outlines the various compartments of peripheral lymphoid organs by characteristic labeling patterns (no such compartments are found in central lymphoid organs). Furthermore ER-TR7 delineates various types of connective tissue compartments in nonlymphoid organs.
The antibody ER-TR7 detects reticular fibroblasts, which constitute the cellular framework of lymphoid and nonlymphoid organs and their products. ER-TR7 is useful to clearly delineated the follicles, periarteriolar lymphoid sheath and marginal zone; the major white pulp compartments. Furthermore in lymph nodes, the capsule, sinuses, follicles, paracortex and medullary cords are clearly delineated.
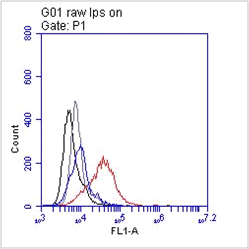
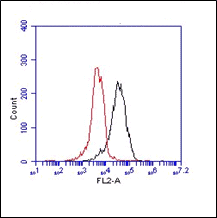
FC: Splenocytes were incubated with ER-TR7 for 30'(Ref.4).
IF: Acetone or PFA fixed cells were quenched with 50mM NH4Cl for 30', blocked and permeabilized with 1.5% goat serum/0.1% saponin in PBS for 45' at RT. Incubation of ER-TR-7 in block and perm solution for 45' at RT. Specific staining was detected with a fluorescent conjugated oat anti-rat IgG (Ref.5).
P: Formalin fixed paraffin sections were deparafined, hydrated in ethanol and stained with ER-TR7 for 3';at RT (Ref.3).