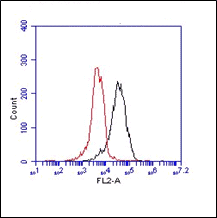
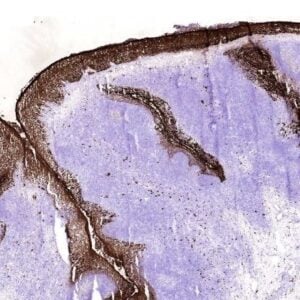
HMGB1 (High Mobility Group Box-1 protein also known as amphoterin) is a highly conserved protein with more than 95% amino acid identity between rodent and human HMGB1. It is a prevalent non-histone chromatin component and a non-sequence specific DNA binding protein. HMGB1 consists of two homologous HMG boxes rich in basic amino acids and an acidic tail at the carboxy-terminus. HMGB1 is involved in the regulation of chromatin structure as well as being involved, either as positive or negative factors with various aspects of DNA replication, transcription, repair, and ligation. HMGB1, identified as a membrane associated protein termed “amphoterin,” mediates neurite outgrowth, tumor outgrowth, and metastasis. It participates in plasminogen activation and is recognized as a late mediator of endotoxin lethality in mice. The monoclonal antibody anti HMGB1, clone 5H6, can be used for Western blot, Flow cytometry and Immunofluorescence.
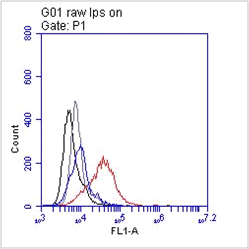
IF: NIH-3T3 cells were fixed with 2% PFA (15 min). After fixation, cells were washed with PBS and incubated for 5 min at 4ºC with permeabilization buffer . The cells were blocked with 1% BSA/10% goat serum in PBS for 10 min and were incubated with HMGB1 mAb 5H6 (10 µg/ml) overnight at 4ºC. The secondary antibody was biotinylated goat anti-hamster IgG at a 1:500 dilution for 1 hour. Finally, streptavidin was used at a 1:500 dilution for 30 min.
W: A band is expected at ~30 kDa using reducing conditions. For non-reducing conditions a band is expected at both 27 kDa and 30 kDa.